Abbott Lab Missteps: Quality Control Issues and Their Potential Impact
An inside look at quality control concerns at an Abbott lab, examining the implications for product safety and consumer trust. We analyze the situation and discuss the future outlook.
Abbott Under Scrutiny: Email Reveals Potential Quality Control Issues
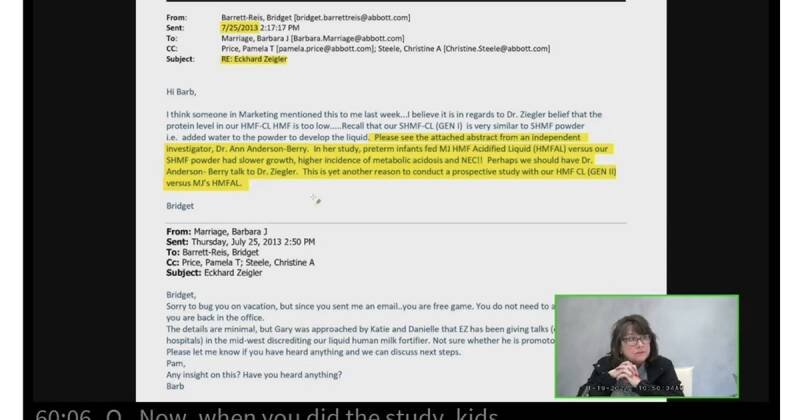
A recent email from Abbott scientist Bridget Barrett-Reis has raised concerns about potential quality control problems within one of the company's laboratories. The email, sent to colleagues, detailed concerning results. The details of the results were not included in the source article, however the use of two exclamation points suggests urgency and concern about the data.
What We Know So Far
While the specific details of the results mentioned in the email remain unclear, the fact that a scientist felt compelled to share them with colleagues suggests a significant deviation from expected standards. The email immediately raises questions about the consistency and reliability of testing procedures within the involved Abbott lab.
Why This News Matters
Quality control in medical labs is paramount. Errors, even seemingly small ones, can have serious consequences. Imagine diagnostic tests giving false results, leading to misdiagnosis, delayed treatment, or unnecessary medical interventions. This could impact patient health, public trust in the healthcare system, and the reputation of Abbott Laboratories.
For example, inaccurate readings in diagnostic tests for infectious diseases could lead to outbreaks. False positives in cancer screenings can cause immense anxiety and potentially lead to unnecessary and invasive procedures. Therefore, maintaining rigorous quality control procedures is crucial for the integrity of the entire healthcare ecosystem.
Our Analysis
In our opinion, this situation warrants a thorough investigation. The mere existence of a concerned email from a scientist indicates a potential breakdown in established protocols. It is crucial to determine:
- What specific tests were affected?
- What was the extent of the deviation from expected results?
- What corrective actions were taken, or are planned?
- Whether the issues are isolated to this particular lab or indicate systemic problems.
This could impact Abbott's reputation. Transparency and a proactive approach to addressing these concerns are essential for rebuilding trust. Any sign of downplaying the issue or a lack of accountability could have serious ramifications for the company's brand image.
Future Outlook
The future hinges on how Abbott responds to these allegations. We anticipate the following:
- Internal Investigation: Abbott will likely conduct an internal investigation to assess the scope and impact of the quality control issues.
- Regulatory Scrutiny: Regulatory bodies, such as the FDA, may launch their own investigations if they deem the situation serious enough. The level of regulatory intervention will depend on the severity and the potential impact of the errors.
- Corrective Actions: Abbott will need to implement corrective actions to address the identified problems. These may include retraining staff, revising protocols, and upgrading equipment.
- Increased Transparency: Abbott will need to be transparent with its findings and corrective actions to reassure the public and regain their trust.
This could impact stock prices. Negative news regarding product quality often affects investor confidence. In our opinion, swift and decisive action is necessary to mitigate any potential financial fallout.
Ultimately, the long-term impact will depend on Abbott's commitment to quality, transparency, and accountability. A proactive and comprehensive approach will be essential to restoring confidence and ensuring the safety of its products. Moving forward, a focus on robust quality assurance measures and continuous improvement is paramount for maintaining its standing in the healthcare industry.